Second law of thermodynamics equation
#SECOND LAW OF THERMODYNAMICS EQUATION FREE#
If you want to skip to any part of this article, feel free to read your interested topic from the table given below.īefore moving further, if you want to know what is thermodynamics, you can refer previous article on basics of thermodynamics. I will also show some practical everyday life examples of the first law of thermodynamics which happens around you in nature. You will get the exact idea about the equation of first law of thermodynamics, it’s examples, applications and limitations too. I will also tell you why it is known as the law of conservation of energy. I’ll explain you this definition/statement of first law of thermodynamics with amazing examples (you will really love this)
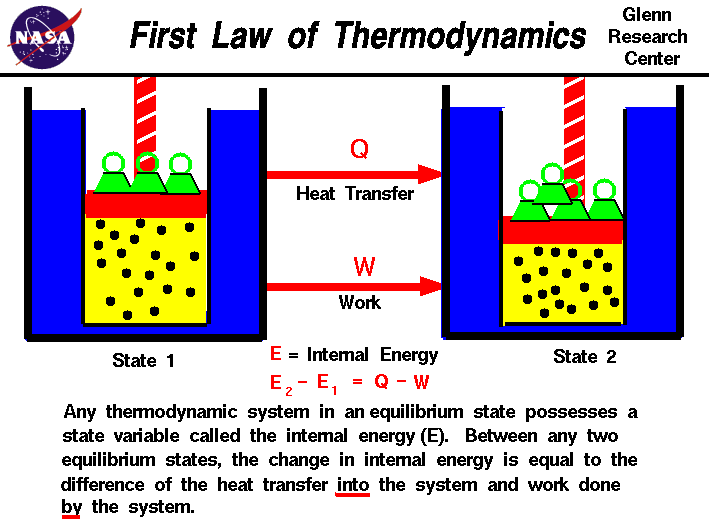
This means that the total amount of energy in the universe remains constant. “Energy can neither be created nor can be destroyed but transformation of one form into another form can be possible.” Now, I’ll show you what is First law of thermodynamics. Total conversion of heat into work is not possible i.e 100% efficiency is not possible as it will lead to a negative change in entropy of universe which is not valid according to the Second Law of Thermodynamics.Here, the heat energy of fuel is converted to mechanical energy. A refrigerator can reduce the temperature to absolute zero Every heat engine has an efficiency of 100%ĭ. The whole of heat can be converted into mechanical energyĬ. The second law of thermodynamics implies :Ī. This causes irreversibilities inside the system and an increase in its entropy.

But if the engine and refrigerator combine together, they form a device that takes up all the heat from the hot body and converts all into work without giving up any amount to the cold body. The engine alone does not violate the second law of thermodynamics. Now suppose an engine working between the same hot and cold bodies takes in heat from hot body converts a part W into work and gives the remaining heat to the cold body. So this is the violation of Clausius statement. Equivalence of Two Statementsįor example, there is a refrigerator that transfers an amount of heat from a cold body to a hot body without having any supply of external energy. It is not at all possible to transfer heat from a cold body to a hot body without the expenditure of work by an external energy source. This means that for obtaining continuous work, a sink is necessary. There is no engine that can convert all the heat taken from the source into work, without giving any heat into the sink. In the heat engine, the working substance takes heat from the hot body, converts a part of it into work and gives the rest to the cold body. It is impossible to convert all the heat extracted from a hot body into work. This is why running an air conditioner for a long period of time, costs you money.

The second law of thermodynamics states that the heat energy cannot transfer from a body at a lower temperature to a body at a higher temperature without the addition of energy. If you ever drop a glass and watch it shatter, you know there is no way of going back in time and getting back the unbroken glass. What is the Second Law of Thermodynamics?
